November 2021 Newsletter
In this Issue…
Notes from Joe
· Show Me The Data!
Legal Update
· 340B Litigation Update: Once more unto the breach, dear friends, once more
Payer Update
· Hemophilia Alliance Network Services (HANS) Update
Alliance Update
· Update on the Alliance HTC Growth Initiative
· 2022 Member Meeting Calendar
Notes from the Community
. Monitoring the Impact of Gene Therapy in Persons with Hemophilia
Show Me The Data!
by Joe Pugliese
As you know, we’ve been working the last few months on production of the DDAVP NS, and I’m excited to share that the second lot was just produced on November 5, and it will have 6 months of dating from the date of manufacture. We’re also making progress on state licensure and expect more states to come online shortly (see map here). We have enjoyed good success with getting payers to reimburse for the product. We still have a ways to go, but our story of ease of use and accessibility coupled with lower costs and great effectiveness for patients is a compelling story. If you have questions or need assistance in getting an insurer to reimburse for the product, please reach out to Jeff Blake – jeff@hemoalliance.org.
As we’ve been working on all of this over the past several months, it became very clear that we needed more data about how big the need was for DDAVP NS. Why is this important? Virtually everyone wants data. In this case, manufacturers want it so they can assess whether they should invest in developing a product. Insurers want to understand how cost effective a particular therapy is. Patients, manufacturers and healthcare providers want to understand how to continuously improve therapies and improve quality of life. The only way to do that is to collect data.
Collecting data is a big business and many private companies have developed infusion logs and worked hard at selling patients and providers on populating their platforms with all manner of data. My concern with this model is that many are private companies, who are looking to build databases that could be sold to the highest bidder – potentially limiting access to and use of that data by the community.
The good news is that we already have an in-house, bleeding disorders community solution to collecting data: ATHN. A 501(c)(3), the ATHN Board is made up of some of the best and brightest clinicians in our community, as well as our federal partners at the NIH, CDC and HRSA. The need for data as well as the need for the data to be used responsibly is only going to become more important. (See article later in the Newsletter on gene therapy)
ATHN has answered the call from the blood disorders community to develop a new tool that enables real-time information sharing from patients and caregivers to the HTC staff. ATHN partnered with four HTCs, the Center for Inherited Blood Disorders, Hemophilia and Thrombosis Center – University of Colorado Anschutz Medical Campus, Newark Beth Israel Medical Center – Children’s Hospital of New Jersey, Indiana Hemophilia Treatment Center, to develop the new Robust Health mobile application.
Robust Health provides a seamless and engaging experience for persons to track treatments, bleeds, Patient Reported Outcome (PRO) measures, and goals. Robust Health also provides users with the ability to visualize their information with interactive views and features two-way text messaging. Users can enable two-way communication between themselves and their HTC via the application.
Robust Health is currently in early release use at the four development partner HTCs through the end of the year. If you are interested in using Robust Health before January 2022, please contact ATHN Support. For more information about Robust Health, visit https://robusthealth.com or contact support@athn.org.
340B Litigation Update: Once more unto the breach, dear friends, once more
by Elizabeth “Issie” Karan and Mike Glomb
Recently, three courts issued opinions related to ongoing 340B Program litigation. If you find yourself confused by headlines describing these outcomes, you are not alone. Before you devote significant brain power to dissecting the intricacies of the courts’ rationale, please note that these decisions likely will be appealed. However, the legal melee (or breach to quote William Shakespeare) does set the stage for congressional action and/or for HRSA to provide clearer guidance on the use of contract pharmacies in the 340B Program.
First, on October 29, 2021, the US District Court for the Southern District of Indiana handed Eli Lilly a mixed outcome. The judge wrote that HRSA “fails to acknowledge or explain the agency’s changed position(s) with regard to its authority to enforce statutory compliance when the alleged violation is entangled with a regulated entity’s failure to comply with the agency’s nonbinding contract pharmacy guidance.” However, the judge also stated that Eli Lily is not permitted to make “unilateral extra statutory restrictions on its offer to sell 340B drugs,” while noting examples of covered entities that lost hundreds to thousands in discounts.
Next, on November 5, 2021, the US District Court for the District of Columbia gave Novartis and United Therapeutics a more favorable opinion stating that drug companies can place conditions on the sales of drugs discounted under the 340B Program to contract pharmacies. However, that same day, the US District Court for the District of New Jersey contradicted this result. In that opinion, the judge told Novo Nordisk and Sanofi that they cannot unilaterally impose restrictions on the 340B Program and that their new policies “must cease.”
Although each suit differs, drug manufacturers are challenging the extent of HRSA’s authority to enforce penalties on drug manufacturers which refuse to provide discounts (or create limitations) on drugs dispensed by covered entities at contract pharmacies. Each case also questions the clarity of HRSA’s contract pharmacy guidance. In large part, the courts agreed that HRSA needs to issue clearer, well-reasoned guidance putting manufacturers on complete notice of the agency’s position for enforcement. As such, all three courts rejected a previously withdrawn HHS Advisory Opinion from December 30, 2020, and vacated letters sent to manufacturers in May 2021, threatening enforcement action. Predictably, both covered entities and drug manufacturers are claiming victory by pointing to different opinions and rationales.
The Hemophilia Alliance team will keep members informed as litigation progresses.
Hemophilia Alliance Network Services (HANS) Update
by Jeff Blake
Exciting News – We have signed our 5th HANS Payer Agreement with Centivo, a national Third-Party Administrator (TPA) with clients throughout the United States. We are working with Centivo to implement our HANS Agreement and we will be contacting HTCs that have signed our HANS PPO Agreement once the implementation is finalized.
We are also very close to signing our 6th HANS Payer Agreement with AultCare, an Ohio based insurance company. We expect to have the Agreement finalized in the next 60 days and will be contacting HTCs in AultCare’s service area that do not currently have an AultCare Agreement.
We are currently working with member HTCs on more than 50 opportunities that represent more than 65,000,000 units of clotting factor and Hemlibra. We are hopeful to finalize some of these opportunities for January 1, 2022 and throughout the first quarter of 2022. Since 2018, we have worked with Payers and our member HTCs to bring back over 40,000,000 units of clotting factor and Hemlibra to our members’ pharmacy programs.
If you would like to learn more and discuss how the Hemophilia Alliance can assist you with your payer challenges and issues, please contact a member of the Hemophilia Alliance Team.
- Update on the Alliance HTC Growth Initiative
by Karen BoweThe Hemophilia Alliance continues to explore ideas to ensure sustainability and growth of member HTC pharmacy programs. Earlier this month, we’ve launched a program focusing on HTCs that dispense less than 5M units/year. We are bringing together staff and physicians from centers across the country to discuss and share topics of mutual interest. Through an open forum format, we are seeking to identify obstacles preventing growth and potential ways to overcome these obstacles, then explore how the Hemophilia Alliance may be able to assist in growing these pharmacy programs.
- “As a participant in the Hemophilia Alliance HTC Growth Initiative, I have already gained some valuable insight after only one meeting with this esteemed group! I feel fortunate to have been asked to participate and look forward to sharing and learning about the challenges of HTCs across the nation and how to implement solutions for growth and sustainability, while keeping our patients at the center of all we do! Networking with other HTC teams is extremely important and I feel that this initiative will provide a forum for participating HTCs to share their challenges and barriers and hopefully gain some resolution. At a minimum, this initiative will set the stage for creating new acquaintances, new partnerships, and provide additional resources for members within the Hemophilia network.”
-
- Louise M. Baca MSN RN
Senior Director, Oncology
Maine Medical Center Cancer Institute - Louise M. Baca MSN RN
- “I am excited to network with other HTCs with similar challenges of being a small, stand alone center. I hope the group will be helpful to leverage our collective strengths to solve these challenges.”
-
- Alison Bartko B.S. Pharm., R.Ph.
Director of Pharmacy
Hemostasis and Thrombosis Center of Nevada - Alison Bartko B.S. Pharm., R.Ph.
The commitment for the group entails 1-hour virtual meetings once a month through April, culminating with an in-person meeting in May, where we will share outcomes of our monthly discussions and offer recommendations to help centers grow their pharmacy programs. We’ll also offer unique perspectives from physicians on barriers and opportunities to grow. If you are interested in joining our group, please reach out to your primary Hemophilia Alliance contact for more information (see map below).
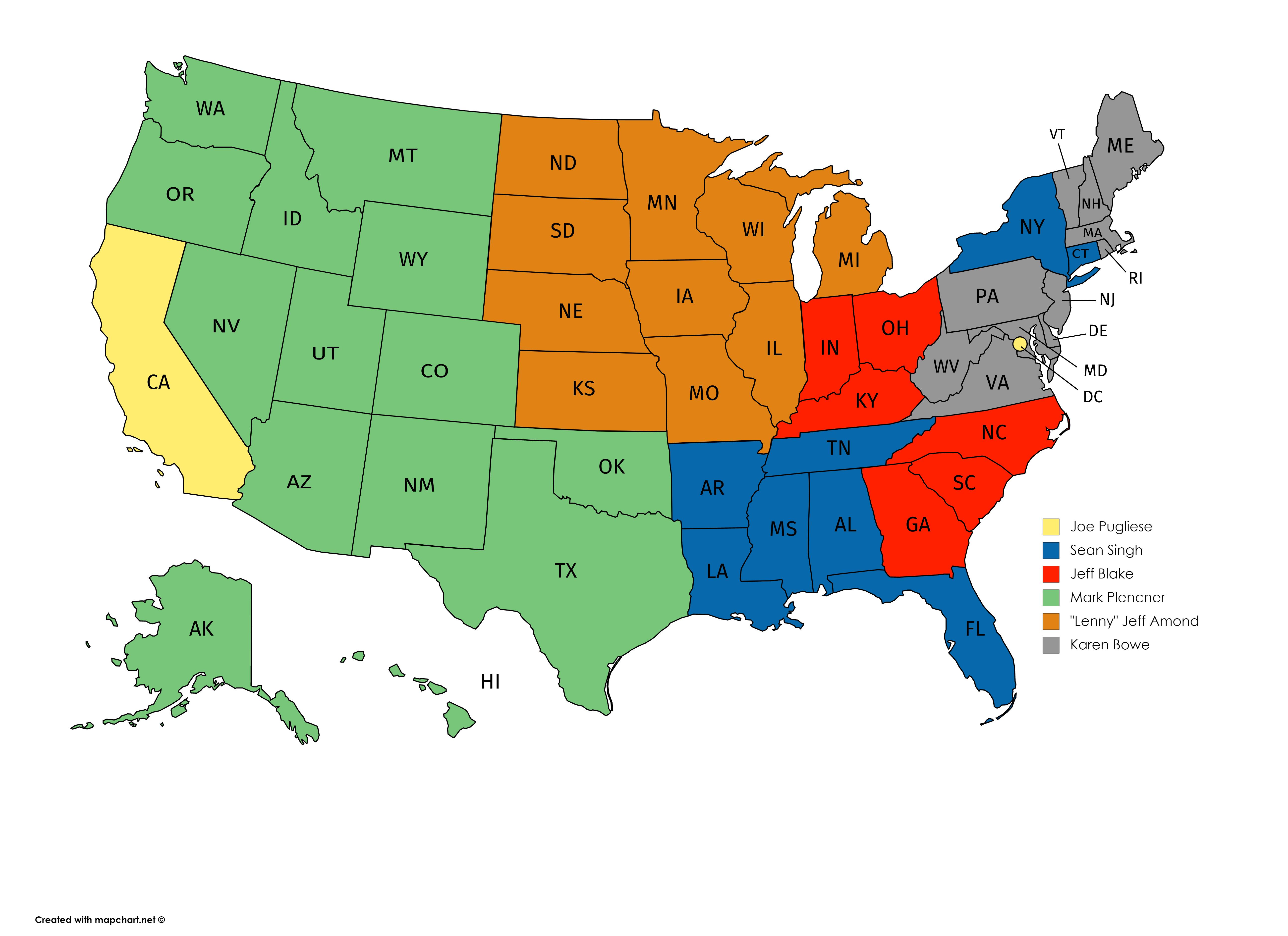
- 2022 Spring Meeting Schedule
by Sean SinghThese meetings will be in-person knowing that we might have to make changes to an alternate format.
Event Name Date Registration Link Board Retreat (Board Members and HA Staff Only) January 9th to 11th Pharmacists CE Conference January 27th to 28th Register Here! New HTC Staff Meeting February 6th to 8th Register Here! Linda Gammage Social Worker Conference February 23rd to 25th Spring members Meeting April 3rd to 5th Hill Day TBD HTC Growth Initiative Meeting May 15th to 17th
Monitoring the Impact of Gene Therapy in Persons with Hemophilia
by Crystal Watson, President and CEO of ATHN
Gene therapy carries the potential for a functional cure of hemophilia. As we move towards the reality of commercial gene therapy treatments for people with hemophilia, it is incumbent upon the community to work together to monitor the impacts of this irreversible one-time therapy. There are many questions that need to be answered. What are the short and long-term safety profiles of these treatments? Why is clinical response, as measured by factor activity level, so variable? Why is durability of factor expression so variable? In order to address the gaps in current hemophilia gene therapy knowledge, data collection and surveillance must be a shared responsibility. Providers and persons with hemophilia receiving the treatment will need to work together to collect standardized data over a lifetime. Long-term data collection is essential in assisting regulators, manufacturers, clinicians, payers, and people with hemophilia and their families in monitoring the safety and effectiveness of these treatments.
Long-term collection of real-world safety and effectiveness data for people receiving hemophilia gene therapy should be considered standard of care. In the United States, this goal can best be supported by leveraging the established collaboration with ATHN and its Hemophilia Gene Therapy Outcomes Arm. The Hemophilia Gene Therapy Outcomes Arm is part ATHN Transcends Study which is rolling out across Hemophilia Treatment Centers (HTCs) in the United States. It is a pragmatic study designed to understand the outcomes of gene therapy with real-world practices across a wide range of products, including the safety of adeno-associated viral vector or lentiviral vector-mediated factor VIII and factor IX therapies when used for participants with hemophilia.
The Gene Therapy Outcomes Arm study aims to enroll all people with hemophilia A or B of any severity, with or without inhibitors, who have received or will receive a gene transfer product within 6 months. Data for the study is to be collected from participants at the time of enrollment and at the following timepoints relative to vector infusion: 3 months, 6 months, 1 year, 18 months, 2 years, and annually thereafter. Participants will be followed longitudinally for at least 15 years after vector infusion. Safety will be measured according to medical events established in the European Haemophilia Safety Surveillance System (EUHASS). In addition to the EUHASS defined events, the study will collect data for adverse events of special interest such as liver toxicity. ATHN will leverage its partner central laboratory organization, Versiti, Blood Center of Wisconsin, to perform factor and inhibitor assays and genetic testing for those who have not previously been tested as part of other projects.
The approach of a central study utilizing a central IRB and a central laboratory is designed to reduce the operational burden on HTCs. In the past it has been the common practice of each manufacturer to establish their own independent registries as products go to market. In our discussions with HTC partners, they have made it clear that this approach is no longer feasible as staffing levels are strained and institutions have implemented stricter controls on which studies can be opened due to the pandemic. In addition, this decentralized approach by manufacturers is unlikely to facilitate a global view of effectiveness.
As clearly articulated by the NHF Medical and Scientific Advisory Council, a global reach will be required to ensure a large enough participant pool to allow for robust evaluation and detection of low-incident safety events. To this end, ATHN has partnered with the international World Federation of Hemophilia Gene Therapy Registry (WFHGTR) to harmonize data collection between the Hemophilia Gene Therapy Outcomes Arm and the WFHGTR to support this global effort. ATHN will serve as the sole source of data from the United States to the WFHGTR. This will help minimize the data collection burden on HTC staff and facilitate the contribution of data from persons with hemophilia to this important global effort. ATHN looks forward to supporting our blood disorder partners as the reality of having commercial gene therapy products has finally arrived at our doorsteps. Stay tuned for more information as the Gene Therapy Outcomes Arm study gets ramped up at treatment centers. We will be sure to share the relevant data and the findings with the community.
Please contact support@athn.org for more information.
Team Alliance Contact Information
We work for you! Please don’t hesitate to contact any of us with any questions or concerns:
| Name | Phone | |
|---|---|---|
| Jeff Blake | jeff@hemoalliance.org | 317-657-5913 |
| Sean Singh | sean@hemoalliance.org | 813-748-7226 |
| Jennifer Borrillo, MSW, LCSW, MBA | borrillo@hemoalliance.org | 504-376-5282 |
| Jeff Amond | amond@hemoalliance.org | 608-206-3132 |
| Jennifer Anders | jennifer@hemoalliance.org | 954-218-8509 |
| Angela Blue, MBA | angela@hemoalliance.org | 651-308-3902 |
| Karen Bowe-Hause | karen@hemoalliance.org | 717-571-0266 |
| Zack Duffy | zack@hemoalliance.org | 503-804-2581 |
| Michael B. Glomb | MGlomb@ftlf.com | 202-466-8960 |
| Johanna Gray, MPA | jgray@artemispolicygroup.com | 703-304-8111 |
| Kiet Huynh | kiet@hemoalliance.org | 917-362-1382 |
| Elizabeth Karan | elizabeth@karanlegalgroup.com | 612-202-3240 |
| Kollet Koulianos, MBA | kollet@hemoalliance.org | 309-397-8431 |
| Roland P. Lamy, Jr. | roland@hemoalliance.org | 603-491-0853 |
| Dr. George L. Oestreich, Pharm.D., MPA | george@gloetal.com | 573-230-7075 |
| Theresa Parker | theresa@hemoalliance.org | 727-688-2568 |
| Mark Plencner | mark@hemoalliance.org | 701-318-2910 |
| Ellen Riker | eriker@artemispolicygroup.com | 202-257-6670 |

Comments are closed.